|
|
|
|
|
 |
|
 |
|
Division of Physiology and Biochemistry
The work on ovine physiology and reproduction was initiated two years after the inception of the institute in the year 1962 but the impetus on research was given after the establishment of the Physiology Section in 1965. At the onset the main thrust of research was on environmental physiology and applied reproduction which was further enlarged in 1975 by up gradation of the section to the status of the division. The division was subsequently renamed in February 2003 as the Division of Physiology and Biochemistry after merger of Biochemistry Section with the division. The major programmes were undertaken on ram semen preservation, artificial insemination and adjudging the physiological adaptability of different strains developed at the institute. During the IX and X plan periods the main emphasis on research was on developing advanced reproductive technologies pertaining to cryopreservation of ram semen, embryo technology and control breeding in ewes while the work on adaptation of sheep to hot semi-arid environment was geared up. The division has successfully completed 1 ICAR ad hoc scheme, 4 NATP and 1 DBT sponsored externally funded research projects. Currently, 1 network progamme, 1 DBT, 1 NICRA sponsored and 1 NFSBFARA are being carried out under external funding projects of the division. The division is credited to report for the first time in the country the birth of lamb following transfer of frozen-thawed embryo in 1998 and IVM-IVF-IVC produced embryo in 1999. The scientist of this division have also been successful in producing 240 elite lambs through ETT and cryoconserving 263 in vivo produced embryos from various sheep breeds.
The division has successfully developed number of protocols suitable to Indian breeds of sheep. The notable research contribution was on development of techniques pertaining to cost-effective indigenous progesterone impregnated intra-vaginal sponges, laparoscope aided embryo transfer and intrauterine artificial insemination, superovulation, ram semen freezing and transcervical artificial insemination. The two technologies namely, ‘Artificial insemination of sheep using short-term preserved ram semen’ and ‘Estrus induction and synchronization in sheep’ have been successfully demonstrated and transferred in the field. The kit developed for ‘Estrus synchronization in sheep and goats’ has also been commercialized.
The scientist of this division have filed two patents through ICAR and conferred with prestigious awards from ICAR, ISCA, NAAS and other scientific societies in national and international seminars. The ICAR Award for Outstanding Team Research was awarded on the first occasion in the biennium 1997-98 for contribution in the field of Small Ruminant Production and on the second occasion in the biennium 2003-04 for contribution in the field of Genetic Improvement of Sheep. The division is credited with several publications in the form of research papers in referred national and international journals, popular articles, book chapters, lead/contributory papers in seminars, research/extension bulletins and manual/teaching aids. The two research contributions of the division have been published as records in the Limca Book of Records.
Salient Research Achievements
- A freezing protocol based on total control of cooling and freezing rates was developed for ex-situ conservation of sheep breed in the form of spermatozoa.
- Non-invasive Transcervical Artificial Insemination Technique was developed and refined for utilizing frozen ram semen.
- Validation of computer-aided semen analysis (CASA) technique for assessment of motion characteristics of ram spermatozoa
- The influence of FecB gene introgression on sperm production and quality was reported for the first time.
- First time reported the influence of age on CASA derived sperm motion characteristics of ram lambs raised under intensive management system in a semi-arid tropical environment.
- Use of Gonadotrophin (FSH, PMSG) and Gonadotrophin releasing hormone (GnRH) in various combinations was successful in inducing multiple ovulations in native sheep. The estrus response ranged from 88.8 to 100 per cent.
- Multiple ovulations were observed in ewes immunized with 30 mer inhibin peptide and its effect persisted profoundly for 3 years as exhibited in terms of 5.3 ovulation rate.
- Recovery of 24 in vivo produced Garole embryos of transferable quality in a single flush was reported. This is the highest recovery for any embryo donor sheep breed reared under tropical environment.
- Garole lambs born of significantly higher birth weight were reported for the first time in the world on transfer of Garole embryos in large size non-prolific recipient ewes.
- Birth of first lamb in the country was achieved from frozen sheep embryo.
- Full-term lamb was produced for the first time in the country from embryo derived from IVM-IVF-IVC techniques.
- Developed heat stress module and nutritional stress module in sheep.
- Thermal stress has an adverse effect on physiological responses, blood parameters and reproductive efficiency of ewes.
- Body condition score of 3-3.5 of ewes is optimum at the time of mating and during gestation for achieving higher lambing and birth weight of lambs.
- Awassi and Patanwadi crossbred lambs were produced in farmers’ flocks through artificial insemination technique using liquid semen.
Future Research Priorities
- Production of 3 lamb crops in 2 years through physiological and reproductive manipulations
- Induction of puberty through administration of kisspeptin
- Improvement in semen processing and preservation
- Fixed time AI with short-term preserved semen
- Studying the effect of heat and nutritional stresses on physiological responses, ovarian functionand in vivo production of embryos and fertility in sheep
- Assessing Resilience of Small Ruminant Production under Changing Climatic Condition in Semi-arid Zone
- Molecular identification and characterization of melatonin receptors gene in relation to reproduction
| Photographs |

Oestrus synchronization kit having sponges, speculum and plunger |
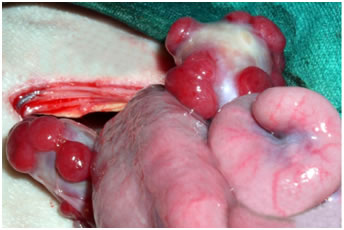 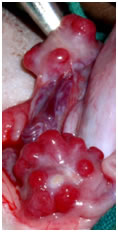 View of corpus luteum on the sheep ovary View of corpus luteum on the sheep ovary |
| |
|

In vivo produced transferable sheep embryos |
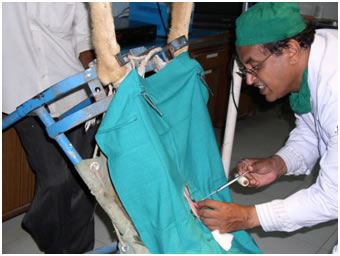
Transfer of embryos by tom-cat catheter into lumen of horn |
| |
|

A - Large size Awassi x Malpura recipient ewe and small size Garole donor ewes
B - Garole rams produced after in vivo derived embryo transfer |
|
| |
|
  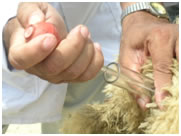
Semen collection, evaluation and AI of sheep with liquid semen |
| |
|

Ram semen freezing using programmable cell freezer |
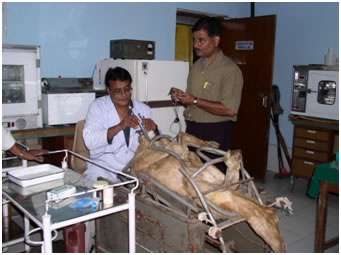
TCAI in sheep |
| |
|
|
|
|
 |
|
 |
|
|
|
|
|


